Abstract
Feed constitutes about 60–70 percent of the total production costs of poultry meat, of which about 175% are animal protein. The protein content of a feed ingredient is a factor of great importance. It has been reported that the protein content of Eisenia fetida differs with the treatment used and it ranges between 50.1% and 66.2%. E. fetida contains 4.95–5.70 g/100 g 20 out of 24 major amino acids, including the ten essential amino acids. The fat content of worm meal has been reported to range from 5–20% of dry matter. A number of mono and polyunsaturated fatty acids are in fairly high concentration in worm meal and the total fatty acids is about 6.6 to 10.5 mg/g. It contains an adequate mineral content and an excellent range of vitamins which are a valuable component of poultry feed. It has been found that it encompasses essential elements such as calcium (1020–7070 μg/g) and iron (1050–2990 μg/g), which are 10 times higher than soya bean and fish meal. Earthworm Eisenia fetida has been found to be a promising alternative source of protein due to its high protein content, amino acid and fatty acid profiles. In this review, we outline the E. fetida characteristics, nutrient composition, processing methods and its effects as a source of feed for chickens.
Similar content being viewed by others
Introduction
Protein can be obtained from many feedstuffs such as meat and fish meals, cereal grains and legume by-products such as soybean meal. Usually, fish meal is commonly used as the most popular protein source for poultry production, and it is frequently the preferred source of protein because of its balance of essential amino acids (Advisory Committee on Animal Feeding Stuffs 2001). Nevertheless, the fish meal has a nutritional constraint in its usage, for example, the high content of histamine that can cause a defect on the gizzard of the poultry (Miculec et al. 2004). The fish meal is also known to cause tainting of chicken meat depending on quantities used and stages of feeding (Swick 1999). Feed cost has constituted about 60–70% of poultry production (Hinrichs and Steinfield 2007; Tiroesele and Moreki 2012). In the least cost feed formulation, the protein content of feed ingredient is the most expensive feed ingredient because conventional feeds are scarce and limited. Though, nutritionists choose ingredients that are high in protein to meet the nutrient requirements of chickens of different categories (Bhatti et al. 2002).
Fishmeal and soybean have been used worldwide as a source of protein for poultry. They are the conventional sources of animal protein in the poultry industry. They have been treasured for their balanced amino acids, and palatability (Davis et al. 2005; Antolovic et al. 2012). Due to the rise in prices of high-quality conventional animal feed required for poultry and competition for feed-in human diets as well as animal husbandry, these feedstuffs are becoming limited and scarce.
Since there is a restriction on the availability of conventional feed for chickens, researchers have come up with alternative protein sources of poultry feed. These alternatives are able to supply adequate essential and non-essential amino acids to enable their synthesis. Moreover, it has been reported that the possible use of some alternative animal protein feedstuff to substitute fishmeal such as earthworms can be a solution (Istiqomah et al. 2009; Ogello et al. 2014).
Earthworms Eisenia fetida have been found to be a good source of protein (Korstecka and Pączka 2006; Sogbesan and Ugwumba 2008). Earthworms with an important high protein component are used to feed chickens, pigs, rabbits, and as a dietary supplement for fish species (Kostecka and Pączka 2006; Stafford and Tacon 1985; Sabine 1986; Mason et al. 1992). The nutritive potential and utilization of earthworm E. fetida as poultry feed ingredients have not been documented in South Africa. Therefore, the objective of this review is to create attentiveness on the nutritional value of E. fetida as a source of protein for poultry.
Description of Eisenia fetida
Eisenia fetida worm belongs to the family of lumbericidae and genus Eisenia, which is known by other names such as the tiger worm, garlic worm, flatworm, cadillac worms and worm for fishing bait (Fadaee 2012). These worms are red, purple or brown in colour and yellowish in their abdomen. The number of its segment is about 80–110 and it is between 23–130 mm in length (Fig. 1). During puberty, the genital belt reaches to 7–9 pieces in between parts 24, 25, 26 or 32.
Eisenia fetida image (Schuld 2004)
Growth of Eisenia fetida
Eisenia fetida growth depends on population density and food rationing (Singh et al. 2013). Their weight increases faster with a decrease in the population density (Garg et al. 2005). On the other hand, Ton et al. (2009) observed that the growth rate of earthworms depends heavily on the type and quality of the substrate. Normally, the growth cycle of worm E. fetida takes 40–60 days for the juvenile to develop into a mature or adult worm (Fadaee 2012). Adult worm weight is approximately 1.5 g at 50 to 55 days. After coming out of the cocoon they are able to reproduce. Adult worms can create a cocoon every three days on average and after 23 days, one- third of newborns come out of the cocoon (Fig. 2). For optimum growth of worms, they require temperatures that range between 15 to 20 ˚C, moisture content from 89 to 90%, oxygen, ammonia content of waste lower than 0.5 mg/g, salt content that is less than 0.5% and pH that ranges from > 5 and < 9 (Sherman 2003).
Growth cycle of earthworm Eisenia fetida (Venter and Reinecke 1988)
Reproduction of Eisenia fetida
Eisenia fetida is hermaphroditic which means that each worm has both female and male reproductive organs (Dynes 2003). Eisenia fetida can reproduce by both sexual and asexual reproduction. They have a high reproduction rate due to their hermaphrodite nature, which allows each individual to produce 9 cocoons, each containing up to 20 eggs at least every 14 days (Musyoka et al. 2019; Tripathi and Bhardwaj 2004). The majority of the species reproduce by cross-fertilisation, although some reproduce cocoon parthenogenetically. In sexual reproduction, worms mate by joining their citellums together with the heads pointing in opposite directions. After copulation and long after the worms separate each worm secretes three eggs or their cocoon from citellim. It takes approximately four days for the formation of cocoon after mating has occurred (Reinecke and Alberts 1988). Incubation lasts for about 23 days for worms to hatch (Fig. 3). Three hatchlings are produced from each cocoon and it takes 20 to 40 days for mature worms to develop citelli (Reinecke and Alberts 1988). According to Edwards and Bohlen (1996), a cocoon takes about 3 to 5 months to hatch.
Lifecycle/reproduction cycle of the earthworm Eisenia fetida (Venter and Reineck 1988)
Feeding earthworms
Earthworms feed on organic matter (Yadav and Garg 2009), including animal waste, plant waste, and urban waste. Animal manure is the main source of feed for E. fetida (Gunadi and Edward 2003). This was also substantiated by Sherman (2003) who specified that cattle horse, rabbit, swine, dairy and beef manure are excellent feed for earthworms, but poultry manure is not recommended because of its high protein and mineral content. It has been found by many authors that they grow faster in cattle manure (Atiyeh et al. 2000; Nagavallmma et al. 2004; Singh et al. 2013). Cattle dung has lower ammonia levels (Ton et al. 2009) and it is rich in nitrogen which enhances the rapid growth of E. fetida (Sharma et al. 2005). They are usually found in areas rich in organic matter, such as the upper topsoil layer, in the forest under piles of leaves or decaying logs, or in piles of manure.
Eisenia fetida nutrient composition
Protein Content of Eisenia fetida
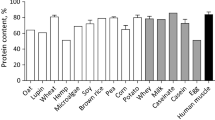
Eisenia fetida is rich in crude protein as shown in Table 1. Crude protein of E. fetida differs with treatments applied on it (Table 1) Eisenia fetida are mostly featured by their high protein content that ranges between 58 and 71% dry weight (Zhenjun et al. 1997; Tiroesele and Moreki 2012). Eisenia fetida has been found to have the highest crude protein content than other earthworm species (Tiroesele and Moreki 2012). Eisenia fetida has 66.8% of crude protein while Eudrilus eugenia (E. eugenia) has 58.38% and Perionyx excavates (P. excavates) has 61.68%. This differs with the study of Sogbesan and Ugwamba (2008) who found that the crude protein for Hyalophora euryalus (H. euryaulis) is lower than 69.8% for P. excavates (Guerrero 1983) and 67.68% for E. fetida (Reinecke and Alberts 1987) but higher than 56.4% and 61.8 for E. fetida reported by Tacon (1994). This variation of protein content may be attributed to the different mediums used to grow earthworms.
Amino acid content of Eisenia fetida
Eisenia fetida has been found to have high amino acid values as compared to other earthworm species (Tiroesele and Moreki 2012). It is better quality in all quantities of the amino acid as shown in Table 2. Earthworm protein is high in essential amino acids compared to other common feeds (Zhenjun et al. 1997). Eisenia fetida contains 20 out of 24 major amino acids, including the ten essential amino acids (Albarran 1996). Segovia (1996) reported that the content of amino acid may be practically 6% of the worm’s dry weight hence; E. fetida is one of the best-known sources of amino acids. Eisenia fetida is rich in lysine which is limiting in many feed stuff and methionine and isoleucine (Albarran 1996; Anitha and Jayraaj 2012). Moreover, it has been also reported by Gabriel et al. (2010) that earthworms have an amino acid composition which is very similar to that of fishmeal and potentially superior to meat meal and contains essential amino acids as phenylalanine, leucine, lysine, methionine and valine (Gabriel et al. 2010).
Fatty acid profile of Eisenia fetida
Eisenia fetida contains appropriate levels of desirable fatty acids and a high amount of omega 3 that distinguish it from other non-conventional feeds (Fadaee 2012). The fat content of worm meal has been reported to range from 5–20% of dry matter (Edward 1988). A number of mono and polyunsaturated fatty acids are in fairly high concentration in worm meal (Table 3). These fats are of special interest of nutrition benefits to poultry feed.
Minerals and vitamins of Eisenia fetida
Eisenia fetida contains an adequate mineral content as shown in Table 4, and an excellent range of vitamins which are a valuable component of poultry feed (Vielma et al. 2001). Essential elements such as Copper (Cu), Manganese (Mn) and Zinc (Zn) are about one to six times higher in earthworm meal than in soybean and fishmeal (Zhenjun et al. 1997). Furthermore, Zhenjun et al. (1997) have reported that the iron content of earthworm fluid is ten times than that of soybean and fishmeal. This feature of earthworm fluid could be exploited to produce special iron (Fe) supplementation for poultry feed production (Zhenjun et al. 1997). Earthworm body is rich in vitamin A and vitamin B compounds 0.1 mg vitamin A, 0.25 mg vitamin B1 and 2.3 mg vitamin B2 per 100 g. It can be concluded that the earthworm E. fetida can be used for mineral and vitamin supplementation in addition to protein supplementation, in poultry feed.
Eisenia fetida as a source of feed for chickens
Eisenia fetida are natural feed for chickens. They can be used as an alternative non-conventional protein source (Bou-Maroun et al. 2013) due to their palatability to chickens (Tiroesele and Moreki 2012). They can be produced and harvested with ease, and they can be produced by simple methods from many kinds of organic material (Tuan and Focken 2009). Several studies have reported that E. fetida are very nutritious for poultry (Barcelo 1988; Reinecke and Viljoen 1991; Istiqomah et al. 2009; Tiroesele and Moreki 2012; Gunya et al. 2016). It is one of the substitutes that can be used instead of fishmeal and soybean because they have a protein content of 64–76% which is greater than the one for fishmeal (45%) and meat meal (51%) (Resnawati 2004). Moreover, it has been reported that the protein content of earthworm meal is close to that of Peru fishmeal and higher than that of Chinese fish meal, hen egg and soybean meal (Dedeke et al. 2013).
The use of E. fetida as a sustainable protein-rich feed ingredient in poultry diet is precisely feasible. They can be reared on low-grade bio-waste and can turn this bio-waste into high-quality protein (Parolin et al. 2020). In addition using earthworm meal as a source of protein for livestock especially for chickens is that they have an excellent chemical composition (Reinbeck et al. 1991), including essential amino acids, especially lysine which is not found in other feed stuff (Anitha and Jayraaj 2012; Tiroesele and Moreki 2012). It has been found that earthworms contain a large amount of protein (64.5—72.9%), essential amino acids (4.95–5.70 g/100 g) (Dedeke et al. 2010) total fatty acids (6.6 to 10.5 mg/g), calcium (1020–7070 μg/g) and iron (1050–2990 μg/g) for chickens. This was also supported by Korstecka and Pączka (2006) and Sogbesan and Ugwumba (2008) who reported that E. fetida is an excellent source of protein, rich in essential amino acids and vitamins. Moreover, they can be used as a substitute for fish meal and soya bean meal, due to their high protein content which ranges between 64–76% (Prayogi 2011). The dietary use of worms has already been analysed for poultry because they are already part of their natural diet. According to Reinecke et al. (1991), E. fetida can be harvested, dried and utilised as a protein source for poultry. There is a contradiction in results found by some researchers regarding earthworm use as poultry feed. Reinecke et al. (1991) found it to be a satisfactory source of protein for growing broilers whereas Koh et al. (1984) reported decreased performance. This difference may be due to the different inclusion levels of earthworm meal used as a protein supplement. The earthworm E. fetida has high-quality protein content along with antimicrobial and antioxidant that could be used as a feed supplement in broiler diet (Rezaeipour et al. 2014). Moreover, it has been reported that it has no anti-nutrient factor which declines performance of broilers (Reinecke et al. 1991). Furthermore, it has been found that growth performance of broilers fed diets containing earthworm meal is equal to that fed fishmeal (Rezaipour et al. 2014). This is in agreement with the observation by Boushy et al. (2000) who found that birds fed on earthworm meal as the major source of protein in the diets have all grown at rates equal to or better than those fed conventional protein meal. Resnawatti (2004) observed that earthworm E. fetida meal in feed up to 5% level has no significant effect on broiler body weight. Dried worm meal included up to 10% in broiler starter diet based on sorghum and soybean meal could be used without negative effects on feed consumption, weight gain and feed efficiency (Ramos-Elonduy et al. 2002). The improvement of body weight of broilers fed with earthworm meal may be attributed to the antibacterial characteristics of earthworm meal (Gunya et al. 2019; Julendra et al. 2012). According to Valembois et al. (1982), the coelomic fluid and the cocoon albumen of the earthworm Eisenia fetida andrei are demonstrated to possess an antibacterial activity. The antibacterial activity and the already known hemolytic activity are due, to the same lipoproteic molecules. The antibacterial activity (bacteriostatic effect) is only directed against the highly pathogenic soil bacteria.
Limitations of Eisenia fetida as poultry diet
Despite their importance, it has been reported that earthworms have limitations as poultry feed. Above 5% worm meal in diets, some researchers have reported a reduction of feed consumption to chickens (Yaqub 1997). Earthworms are the main intermediate of the internal parasite in free range chickens (LeeMaster 2007). Chickens may pick up parasites directly by ingesting earthworms which carry the eggs of internal parasites specifically roundworms and cecal worm nevertheless, processing of works like drying can limit the intake of the parasite. Internal parasites have a negative impact to chickens that can result in poor growth rate and feed efficiency, decrease egg production and even death in severe infections (Gabanakgosi et al. 2012). Earthworm production is economically feasible with high technology rearing methods (Edward and Arancon 2010). However, current methods of harvesting earthworms from organic waste are labour-intensive. This has been regarded as the greatest barrier to the successful commercial production of earthworm protein. Hence, earthworm as a source of protein has not been adopted commercially in developing countries (Edward and Arancon 2010). The earthworm meal is produced at an economical price, and additionally, the value of vermicompost produced can also be taken into account as complimentary income (Edward and Arancon 1994). Therefore, there is a need to come up with most efficient earthworm harvesting methods for the reason that its economic value is extremely valuable as feed for poultry as fishmeal because of its amino acids, fatty acids, minerals and vitamins content (Edward and Arancon 1994).
Conclusion
In conclusion, understanding the description, feeding, growth and reproduction is vital for production of earthworms which can then be used as animal feed. The advantage of utilising E. fetida as chicken feed are that they grow very rapidly, low inputs are required, and they convert organic matter diet into high-quality protein feed. Protein is an important nutrient in poultry diets that cannot be comprised. Earthworm E. fetida had demonstrated a potential and equal protein substitute which can be used as a protein source for poultry diets. In addition, E. fetida has a potential to be used as a protein source to poultry. It has a crude protein content that ranges between 60–70%, which is more than of fish and meat meal currently used in the poultry industry. Besides its good protein, it is better in quality in all amino acids and lysine which is limiting in many feed stuffs. Eisenia fetida contain vitamins and minerals, hence it can also be used as mineral and vitamin supplement. Furthermore, this study provide awareness about benefits of earthworm as a feed source for poultry and its use could play an important role in the sustainable of poultry production.
References
Advisory Committee on Animal Feeding Stuffs (2001) The use of fish meal in animal feeds. Eight Meeting of ACAF- Agenda Item 3
Albarran GN (1996) Formulation de alimentos concentrados para animalers a parti de harina de lombriz (Tesis de lecenciatura. Menda-Venezuela Universidad de Los Andes, Ingeniera) Laboratorio de Ciencia de los Alimentos
Anitha J, Jayraaj IA (2012) Nutritional and oxidant evaluation of earthworm powder. Int J Pharm 3:177–180
Antolovic N, Kozul V, Antovic M, Bolotin J (2012) Effect of partial replacement of fish meal and soya bean meal on the growth of Juvenile Saddled Bream. Turk J Fish Aquat Sci 12:247–252
Atiyeh RM, Dominguez J, Subler S, Edward C (2000) Changes in biochemical properties of cow manure during processing by earthworms (Eisenia andrei, bouche) and the effect on seedling growth. Pedobiologia 44:709–724
Barcelo PM (1988) Production and utilisation of earthworm for broiler in the Philippines. Tropicult 6:21–24
Bhatti BM, Talat T, Sardar R (2002) Estimation of crude fibre and crude protein in commercial poultry rations and some important feed ingredients. Pak Vet J 22:5–7
Bhuvaneshwaran T, Sanjay G, Jayakumar N et al (2019) Potentiality of earthworm as replacement for fish meal and its role in aquaculture. J Aqucult Trop 34:115–127
Bou-Maorun E, Louiac C, Loison A et al (2013) Impact of preparation process on protein structure and the volatile compounds in Eisenia fetida protein powders. Food Nutri Sci 4:1175–1183
Boushy ARY, van der Poel AFB (2000) Poultry feed from waste processing and use. 2nd Ed, Kluwer Academic Publishers, U.S.A, pp. 51–52
Davis DE, Miller CL, Phelps RP (2005) Replacement of fishmeal with soybean meal in the production diets of Juveline Red Snapper. Lutjanus campechanus J World Aqualt Soc 36:114–119
De Chaves RC, Paula RQ, Gücker B et al (2015) An alternative fish feed based on earthworm and fruit meals for tilapia and carp postlarvae. Braz J Biol Sci 13:15–24
Dedeke GA, Owa SO, Olurin KB et al (2013) Partial replacement of fishmeal by earthworm meal (Libyodrilus violaceus) in the diet for 38 African catfish, Clarias gareipinus. Int J Fish Aquac 5:229–233
Dedeke GA, Owa SO, Olurin KB (2010) Amino acid profile of four earthworm species from Nigeria. Agriculture and Biology Journal of North America, ScienceHuβ, http://www.scihub.org/ABJNA
Dynes RA (2003) Earthworms, Technology information to enable the development of earthworm production, Australian Government, Rural Industrial Research, and Development Corporation, Australia
Edward CA (1988) Breakdown of animal, vegetable and industrial organic wastes by earthworm in waste and in the environment. Academic Publishing, Netherlands, SPB
Edward CA, Arancon NQ (2010) The Science of vermiculture: The use of earthworms in organic waste management, The Ohio State University, Columbus, Ohio, U.S.A
Edward CA, Arancon NQ (1994) The use of earthworms in the breakdown of Organic Waste to produce vermicompost and animal feed protein, in:Earthworm Ecology 2nd Edition, CRC Press, U.S.A. pp 345
Edwards CA, Bohlen PJ (1996) Biology and Ecology of Earthworms, 3rd edn. Chapman & Hall, London
Fadaee R (2012) A review on earthworm Eisenia fetida and its implications. Ann Biol Res 3:2500–2506
Gabriel A, Dedeke SO, Kayode BO (2010) Amino Acid profile of four earthworm species from Nigeria. Agric Biol J North Amer 1:97–102
Garg VK, Chand S, Chhilar A, Yadav A (2005) Growth and reproduction of Eisenia Fetida in various Animal wastes during vermicomposting. Appl Ecol Environ Res 3:51–59
Gabanakgosi K, Moreki JC, Nsoso SJ, Tsopito C (2012) Ethnoveterinary medicine usage in family chickens in the selected four villages of Botswana. J Anim Vet Adva 2: 586-594
Guerrero RD (1983) The culture and use of Perionxy excatus as protein resource in the Philippines. In: Satchell JE (ed) Earthworm Ecology. Chapman and Hall, London, pp 309–319
Gunadi B, Edward CA (2003) The growth and fecundity of Eisenia fetida (Savigny) in cattle solid pre-composted for different periods. Pedobiologia 64:15–23
Gunya B, Masika PJ, Hugo A, Muchenje V (2016) Nutrient composition and fatty acid profiles of oven-dried and freeze dried earthworm Eisenia fetida. J Food Nutr Res 4:343–348
Gunya B, Masika MV, PJ, (2019) The potential of Eisenia fetida as a protein source on the growth performance, digestive organs size, bone strength and carcass characteristics of broilers. J Appl Poult Res 28:374–382
Hinrichs J, Steinfeild H (2007) Feed availability inducing a structural change in the poultry sector Thyme, O Pillinf, D., eds. Poultry in the 21st century: avian influenza and beyond. Proc Int Poul ZCOnf, held 5–7 November 2007, Bangkong, Thailand. FAO Animal Production and Health Proceedings, Np. 9. Rome. 2008. Available at https://www.fao.org/againinfo/resources/en/pubs_aprod.html
Istiqomah L, Sofyan A, Diamante E, Julendra H (2009) Amino acid profile of earthworm and earthworm meal (Lumbricus rubellus) for animal feedstuff. J Trop Anim Agric 32:253–257
Julendra H, Damayanti E, Istoqomah L, Nurhayati S, Karimy MF (2012) The effectiveness of earthworm meal supplementation as antibiotic growth promoter replaced with different processing method, proceeding of the 1st Poultry International Symposium, 145–157
Koh TS, Oh CH (1984) Biological values of earthworm (Eisenia fetida) cake protein in chick. Korean J Anim Sci 26:389–395
Kokstecka J, Packza G (2006) Possible uses of earthworm Eisenia fetida biomass for breeding aquarium fish. Eur J Soil Biol 42:5231–5233
LeeMaster BR (2007) Parasite Important to poultry in Hawai’i and their control, Livestock Management, Cooperative Extension Service, College of Tropical Agriculture and Human Resources, University of Hawai’i, Manca.
Mason WT, Rottman RW, Dequine JF (1992) Culture of earthworm for Bait for Fish Food. Publication, #CIR 1053 of the Department of Fisheries and Aquatic Sci., Florida Cooperation Extension Service, Institute of Food Agric. Sci. University of Florida
Miculec Z, Mas N, Masek T, Stmotiae A (2004) Soybean meal and sunflower meal as a substitute for fish meal in broiler diet. Vet Arch 74:271–279
Musyoka SN, Liti DM, Ogella EO et al (2020) Using earthworm, Eisenia fetida, to Bio-convert Agro-industrial wastes for Aquaculture Nutrition. BioRes 15:574–587
Musyoka SN, Liti DM, Waidbacher H (2019) Utilization of the earthworm, Eisenia fetida (Savigny, 1826) as an altanative protein source in fish processing: A review. Aquacult Res 50:2301–2315
Nagavallmma KP, Wani KP, Stephanen L, et al (2004) Vermicomposting: Recycling waste into valuable organic fertiliser, Global Theme on Agrocosystem Report no.8. Patancheru 502 325, Anhara Pradesh, India: International Crop Research Institute for Semi-Arid Tropics
Ogello EO, Munguti JM, Sakakura Y, Hagiwara A (2014) Complete replacement of fish meal in the diet of Nile Tilapia (Oreochromis niloticus) Grow-out with an alternative protein source. A review Int J Adv Res 2:926–978
Parolin M, Ganzaroli A, Baccenetti J (2020) Earthworm as an altanative protein source in poultry and fish farming; current. Department of Agriculture and Evironmental Science. UNiversita degli Studi Milano, Milan, Italia, Availavble at https://core.ac.uk/download/pdf/333583482.pdf
Prayogi HS (2011) The effect of Earthworm meal supplementation in the diet on Qual’s growth performance in an attempt to replace the usage of fish meal. Int J Poult Sci 10:804–806
Ramos-Elorduy J, Gonzalez EA, Hernandez AR et al (2002) Use of tenebrio molito (Coleoptera:tenebrionide) to recycle organic waste and as feed for broiler chickens. J of Econom Entomol 95:214–220
Reinecke AJ, Alberts JN (1988) The chemical and amino acid composition of the compost worm (Eisenia fetida Oligocheata) as potential source of protein for animal feed. S.A
Reinecke AJ, Alberts JN (1987) The chemical and amino acid composition of the compost worm (Eisenia fetida Oligocheata) as potential source of protein for animal feed. S.A. Tydskrif Vir Natuurwetenskap en Tengnologie 6:1–14
Reinecke AJ, Hayes JP, Cilliers SC (1991) Protein quality of three different species of earthworms. S Afr J Anim Sci 21:99–103
Reinecke AJ, Viljoen SA (1991) A comparison of the biology of Eisenia fetida and Eisenia andrei (Oligochaeta). Biol Fertil Soils 11(4):295–300
Resnawati, (2004) Technology in animal husbandry and veterinary as motor of change in animal agribusiness. Puslitbang Peternakan 2:473–478
Rezaeipour V, Nejad OM, Miri HD (2014) Growth performance, blood metabolites and jejunum morphology of broiler chickens fed diets containing earthworm (Eisenia fetida) meal as a source of protein. Int J Adv Biol Biom Res 2:2483–2524
Rufchaei R, Hoseinifar SH, Nedaei S, et al (2019) No-specific immune responses, stress resistance and growth performance of Caspian roach (Rutilus caspicus) fed supplemented with earthworm (Eisenia fetida) extract. Aquaculture, 511
Sabine JR (1986) Earthworms as a source of food and drugs. Earthworm ecology from Darwin to Vermiculture. Chapman and Hall, London, New York, pp 285–296
Schuld M (2004) Lombriz roja (Eisenia foetida). Available at: https://www.researchgate.net/profile/Ana-Carrera-Aguilar/publication/292129975_Que_paso_en_Mexico_durante_la_primera_decada_del_siglo_XXI_Una_perspectiva_socioeconomica_regional/links/56a910bc08aef6e05df28a76/Quepaso-en-Mexico-durante-la-primera-decada-del-siglo-XXI-Una-perspectiva-socioeconomica-regional.pdf#page=106
Segovia E (1996) Analysis flsico-quimico de la harina de lombriz Eisenia fetida (Tesis de licenciatura, Ingeniera). Universidal Agraria La Molina, Lima-Peru
Sharma S, Pradhan K, Satya S, Vesudevan P (2005) Potentiality of earthworms for waste management and in other uses. A review J Amer Sci 1:4–16
Sherman R (2003) Raising Earthworms successfully. North Carolina State University Raleigh, NC, North Carolina Cooperative Extension Service
Singh K, Nath R, Rai R, Shikla RC (2013) Food Preference of Eisenia Fetida among different combinations of animal dung and agro/kitchen waste. Bot Res Int 6:23–26
Sogbesan AO, Ugwumba AAA (2008) Nutrient value of some non-conventional animal protein feedstuffs used as fishmeal supplement in aquaculture practice in Nigeria. Turk J Fish Aquat Sci 8:159–164
Stafford EA, Tacon AGJ (1985) The nutritional evaluation of dried earthworm meal included at low level in production diets for rainbow trout, Salmo Gardner. Richardson Aquacult Fish Manag 16:213–222
Swick RA (1999) Consideration in using protein meals for poultry and swine, ASA Technical Bulletin Vol. AN21–1999
Tacon AGJ (1994) Feed ingredient for carnivore’s fish species. Alternative to fishmeal and other fishery resources. FAO of United Nations, Fisheries Circular No 881. FIRI/C881, Rome
Tiroesele B, Moereki JS (2012) Terminates and Earthworms as a potential alternative source of protein for poultry. Int. J Agro Vet. Med, Sci 6:368–376
Ton VD, Hanh HQ, Linh NG Duy GV (2009) Use of red worm (Perionyx excavates) to manage agricultural waste and supply valuable feed for poultry. Livest Res Rural Dev 21(11)
Tripathi P, Bhardwaj B (2004) Earthworms in waste and environmental management. Bioresour Technol 92:275–283
Tuan NN, Focken U (2009) Earthworm meal as a potential protein source in diets for common carp (Cyprinus carpio L.). Biophy. and Socio-econom. Frame Cond Sust Man Nat Resourc 44:43–58
Valembois P, Roch P, Lasseques M, Davant N (1982) Bacteriostatic activity of a chloragogen cell secretion. Pedobiologia 24:191–19
Venter JM, Reinecke AJ (1988) The life-cycle of the compost worm Eisenia fetida (Oligochacta). S Afri J Zool 23:161–165
Vielma R, Carrero P, Rondón C, Medina A (2001) Contenido de minerales y elementos trazas en la haring de lombriz californiana (Eisenia fetida). 51st Annual Convention of the Venezuelan Association for the Advancement of Science (AsoVAC); (2001) 16–21. National Experimental University of Táchira, Táchira-Venezuela
Vodounnou DS, Juste V, Kpogue DNS, Apollinaire MG, Didier FE (2016) Culture of earthworm (Eisenia fetida), production, nutritive value and utilization of its meal in diet for Parachanna obscura fingerlings reared in captivity. Int J Fish Aquat 4:01–05
Yadav A, Garg VK (2009) Feasibility of nutrient recovery from industrial sludge by vermicomposting technology. J Hazard Mat 168:262–268
Yaqub HB (1997) Earthworm and maggot meal as a potential fish meal replacement, Repository of Ocean Publications, Marine Fisheries Research Division, Tema, Available at http://www.oceandocs.org/handle/1834/1268
Zhenjun S, Xianchun Lihui S, Chunyang S (1997) Earthworm as a protein source. Ecol Food Nutri 36:221–236
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gunya, B., Masika, P.J. Eisenia fetida worm as an alternative source of protein for poultry: a review. Int J Trop Insect Sci 42, 1–8 (2022). https://doi.org/10.1007/s42690-021-00531-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-021-00531-6